Evaluation of corrosion of steel embedded in concrete exposed to carbonation AAS using a factorial experiment with repeated measures.
Main Article Content
Keywords
analysis of variance, repeated measure, alkali activation, carbonation, corrosion.
Abstract
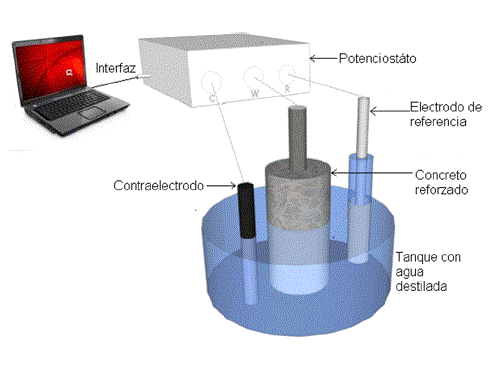
This article presents various techniques for assessing corrosion, the linear polarization resistance and galvanostatic pulse, the analysis was performed using analysis of variance models using a factorial experiment with three factors, one repeated measure, because on the same experimental unit (reinforced concrete), it experienced several samples to be measured in time (0 h=1, 350 h=2, 700 h=3, 1050 h=4, 1700 h=5 y 2600 h=6). This makes the observations are not independent, the other two factors relate to, the binder (ce: 1 = activated slag [AAS] and 2 = ordinary Portland concrete [OPC]) and the exposure condition (ca: 1 = y 2 = environmental exposure accelerated carbonation). These factors were discussed according to the results of the electrochemical properties. Initially performed the statistical processing of each of the variables in relation to the properties and analyzed the interaction between them. Where they established the differences in materials used as coating steel, concrete, OPC and AAS. OPC concrete specimens, exhibited a resting potential, polarization resistance and ohmic (CO2 condition) than those obtained for reinforced concrete in AAS.
PACS: 82.45.Bb
MSC: 62K15
Downloads
References
[2] E. A. Sacco, J. D. Culcasi, C. I. Elsnera, and A. R. D. Sarlia., “Comportamiento en CNS de chapas de acero recubiertas por inmersión con Zn y aleación 55% Al-Zn, deformadas por tracción uniaxial.” in Jornadas SAM Conamet, 2001. 239
[3] D. M. Roy, W. Jiang, and M. R. Silsbee., “Chloride diffusion in ordinary, blended, and alkali-activated cement pastes and its relation to other properties,” Cement and Concrete Research, vol. 30, no. 12, pp. 1879–1884, 2000. 239
[4] J. Maldonado, R. D. Gutierrez, and S. Delvasto, “Propiedades de un subproducto industrial activado alcalinamente,” in VI Congreso Nacional de Corrosión, Bogotá, Colombia, 2001, pp. 1–5. 239
[5] D. Gaspar, “Seminario S.3: patología del hormigón, durabilidad (corrosión). X Curso de Estudios Mayores de la Construccion, Edificación, su Patología y Control de Calidad,” in Cemco 85, Madrid, España, 1985, pp. 205–210. 239
[6] N. I. Fattuhi and B. P. Hughes, “The performance of cement paste and concrete subjected to sulphuric acid attack,” Cement and Concrete Research, vol. 18, no. 1, pp. 545–553, 1988. 239
[7] Saraswathy and S. Ha-Won, “Evaluation of corrosion resistance of portland pozzolana cement and fly ash blended cements in pre-cracked reinforced concrete slabs under accelerated testing conditions,” Materials Chemistry and Physics, vol. 104, no. 2-3, pp. 356–361, 2007. 239
[8] S. Yodmunee and W. Yodsujai, “Study on corrosion of steel bar in fly ash based geopolymer concrete,” in International Conference on Pozzolan, Concrete and Geopolymer, Khon Kaen, Thailand, 2006, pp. 78–85. 240
[9] R. Kuehl, Diseños de experimentos: Principios estadísticos de diseño y análisis de investigación. I.T.P Latin America, México, 2001, no. 9706860487. 240
[10] ASTM, Test Method for Half-Cell Potentials of Uncoated Reinforcing Steel in Concrete, ASTM Standard C876- 09, American Society for Testing and Materials, 2010. 243
[11] P. Fernández, P. Livacic-Rojas, and G. Vallejo, “Cómo elegir la mejor prueba estadística para analizar un diseño de medidas repetidas,” International Journal of Clinical and Health Psyc, vol. 7, no. 1, pp. 153–175, 2007. 244
[12] F. Bellavance, S. Tardif, and M. A. Stephens, “Tests for the analysis of variance of crossover designs with correlated errors,” Biometrics, vol. 52, no. 2, pp. 607–612, 1996. 244
[13] A. R. Borzillo, J. E. Crowley, and J. B. Horton, “Non-ferrous metal coated products and method of production thereof. Bethlehem Steel Corporation,” 1989. 246
[14] A. Bentur, N. Berke, and S. Diamond., Steel Corrosion in Concrete: Fundamentals and civil engineering practice. E & FN Spon, 1997, no. 0419225307. 247
[15] N. M. Ihekwalva, B. B. Hope, and C. M. Hansson, “Pull-out and bond degradation of steel rebarsin ECE. Concrete,” Cement and Concrete Research, vol. 26, no. 2, pp. 267–282, 1996. 250
[16] D. W. Law, J. Cairns, S. G. Millard, and J. H. Bungey, “Measurement of loss of steel from reinforcing bars in concrete using linear polarisation resistance measurements,” NDT&E International, vol. 37, no. 5, pp. 381–388, 2004. 250
[17] S. Sathiyanarayanan, P. Natarajan, K. Saravanan, S. Srinivasan, and G. Venkatachari, “Corrosion monitoring of steel in concrete by galvanostatic pulse technique,” Cement and concrete composites, vol. 28, no. 7, pp. 630–637, 2006. 252
[18] B. Elsener, H. Wojtas, and H. Bohni, “Galvanostatic pulse measurements rapid on-site corrosion monitoring,” Proc. Inter. Conf. Corrosion and Corrosion Protecting of Steel in Concrete, vol. 1, no. 1, pp. 236–246, 1994. 252